GAMP published its first draft guidance in February 1994, with version 1.0 of it arriving in March 1995. Food and Drug Administration expectations for Good Manufacturing Practice (GMP) compliance of manufacturing and related systems. GAMP itself was eventually founded in 1991 (with the previously mentioned Margetts as chairman of the editorial board) to deal with the evolving U.S. A second version was created upon additional feedback.
Gamp software categories software#
This culminated in a five-page document called VMAN I, mapping the older installation qualification (IQ), operational qualification (OQ), and performance qualification (PQ) phases of equipment validation to a more modern software validation lifecycle. They worked with ICI Pharmaceuticals' Tony Margetts on the problem of validating systems that were increasingly becoming more software-based than mechanical- and electrical-based. GAMP's origins can be traced to the United Kingdom in 1988, when software developers David Forrest and Colin Jones, through their company FJ Systems, developed real-time control and production information management control systems for pharmaceutical manufacturers. Categorising software is used to support the approach to validation based on the difficulty and individuality of the computerised system. These documents then form the basis for the traceability matrix and for the formal testing of internal acceptance, factory acceptance, and site acceptance. The process begins with a user requirements specification for the machine, from which a functional requirement and a design specification are created. It is a formal process of thorough documentation, testing, and logical process steps that validate clients' required specifications. In October 2014, Irish tech company Dataworks Ltd. GAMP is largely about automated system validation.

As a result, GAMP covers all aspects of production, from the raw materials, facility, and equipment, to the training and hygiene of staff.

One of the core principles of GAMP is that quality cannot be tested into a batch of product but must be built into each stage of the manufacturing process.
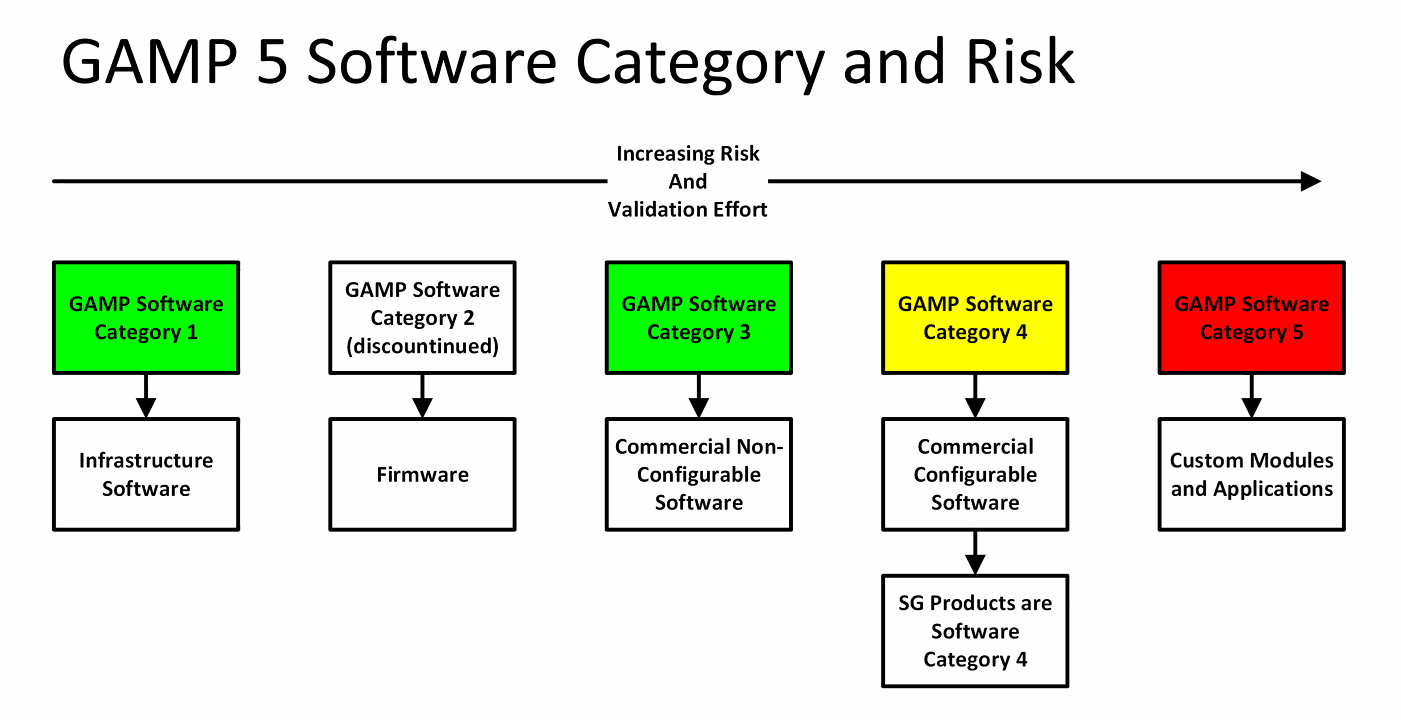
Good Automated Manufacturing Practice ( GAMP) is both a technical subcommittee of the International Society for Pharmaceutical Engineering (ISPE) and a set of guidelines for manufacturers and users of automated systems in the pharmaceutical industry. The area of automated pharmaceutical manufacturing is influenced in part by GAMP and its associated guidelines.
